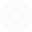
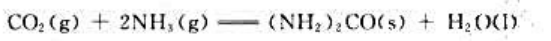题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
[主观题]
已知钡的升华焓=180.0kJ·mol-1,第一、第二电离能分别为507.94kJ·mol-1和971.44kJ·
已知钡的升华焓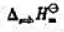 =180.0kJ·mol-1,第一、第二电离能分别为
=180.0kJ·mol-1,第一、第二电离能分别为
507.94kJ·mol-1和971.44kJ·mol-1,Ba2+(aq)的标准摩尔生成烩的相对值(Ba2+,q)=-537.64kJ.mol-1.试用热力学循环计算;(1)Ba2+(g)的标准摩尔生成焓ΔlHθm(Ba2+,g);(2)Ba2+(g)的水合焓ΔhHθm(Ba2+,g).
查看答案
 如果结果不匹配,请 联系老师 获取答案
如果结果不匹配,请 联系老师 获取答案


 更多“已知钡的升华焓=180.0kJ·mol-1,第一、第二电离能…”相关的问题
更多“已知钡的升华焓=180.0kJ·mol-1,第一、第二电离能…”相关的问题
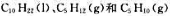 的标准摩尔燃烧焓分别为-6752kJ·mol- 1、-3492kJ·mol-1和-3364kJ·mol-1。计算298.15K裂解反应
的标准摩尔燃烧焓分别为-6752kJ·mol- 1、-3492kJ·mol-1和-3364kJ·mol-1。计算298.15K裂解反应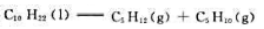 的标准摩尔焓变。
的标准摩尔焓变。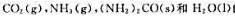 的标准摩尔生成焓和标准摩尔熵如下表所示:
的标准摩尔生成焓和标准摩尔熵如下表所示: