 题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
[单选题]
EDTA配位滴定化学计量点pM'值的计算式是()
A.1/2(pCM(SP)+IgKMY')
B.1/2(pCM(SP)+lgKMY)
C.1/2(pCM(SP)+pKMY')
D.1/2(pKMY'+lgCM(sp))
查看答案
 如果结果不匹配,请 联系老师 获取答案
如果结果不匹配,请 联系老师 获取答案
 题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
A.1/2(pCM(SP)+IgKMY')
B.1/2(pCM(SP)+lgKMY)
C.1/2(pCM(SP)+pKMY')
D.1/2(pKMY'+lgCM(sp))
 如果结果不匹配,请 联系老师 获取答案
如果结果不匹配,请 联系老师 获取答案
 更多“EDTA配位滴定化学计量点pM'值的计算式是()”相关的问题
更多“EDTA配位滴定化学计量点pM'值的计算式是()”相关的问题
A.0%
B.50%
C.100%
D.150%
A.等于7.0
B.小于7.0
C.等于8.0
D.大于7.0
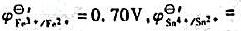 0.14V)
0.14V)
准确量取30.00ml弱酸,加水稀释至100ml。以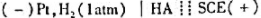 为测定电池,以0.1000mol/LNaOH溶液滴定弱酸HA。当酸被中和一半时,电池电动势为0.524V;化学计量点时,电池电动势为0.749V。φSCE=0.242V。计算:
为测定电池,以0.1000mol/LNaOH溶液滴定弱酸HA。当酸被中和一半时,电池电动势为0.524V;化学计量点时,电池电动势为0.749V。φSCE=0.242V。计算:
(1)弱酸HA的Ka。
(2)原弱酸HA的浓度。
某三元酸H2A的标准解离常数分别为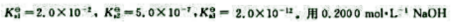 标准溶液滴定H2A溶液至第一化学计量点时,溶液的pH=().可选择()作指示剂。滴定至第二化学计量点时,溶液的pH=()可选择()作指示剂。
标准溶液滴定H2A溶液至第一化学计量点时,溶液的pH=().可选择()作指示剂。滴定至第二化学计量点时,溶液的pH=()可选择()作指示剂。