 题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
[单选题]
移取双氧水2.00L(密度为1.010g/mL)到250m的容量瓶中,并稀释至刻度,吸取25.00mL,酸化后用c(1/5KMnO4)=0.1200m01/L的KMnO4溶液29.28mL滴定至终点,则试样中H2O2的含量为()。M(H2O2)=34.02g/mol。
A.2.96%
B.29.59%
C.5.92%
D.59.17%
查看答案
 如果结果不匹配,请 联系老师 获取答案
如果结果不匹配,请 联系老师 获取答案
 题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
A.2.96%
B.29.59%
C.5.92%
D.59.17%
 如果结果不匹配,请 联系老师 获取答案
如果结果不匹配,请 联系老师 获取答案
 更多“移取双氧水2.00L(密度为1.010g/mL)到250m的…”相关的问题
更多“移取双氧水2.00L(密度为1.010g/mL)到250m的…”相关的问题
A.5.64mL
B.8.33mL
C.10.22mL
D.16.6mL

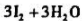 ,析出的碘用Na2S2O3溶液滴定,用去25.10ml。求:Na2S2O3溶液的浓度。(MHNO3=214.0)
,析出的碘用Na2S2O3溶液滴定,用去25.10ml。求:Na2S2O3溶液的浓度。(MHNO3=214.0)
A.4m
B.40m
C.400m
D.60m